In 2016, the BSDB introduced the Cheryll Tickle Medal, which is being awarded annually to a mid-career, female scientist for her outstanding achievements in the field of Developmental Biology. 
The BSDB is proud to announce the 2020 awardee, Tatjana Sauka-Spengler.

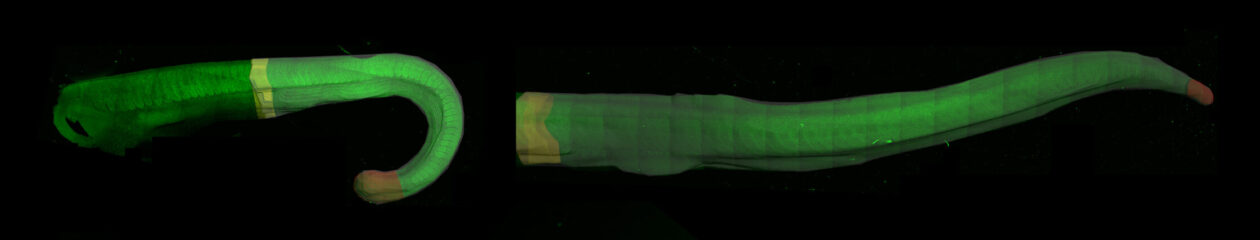
Originally from Bosnia and Herzegovina, Tatjana completed her undergraduate studies in Physics at the University of Sarajevo and soon after sought asylum from the Bosnian War in Czech Republic, where she became a High School teacher at the Gymnazium of Pardubice (1992-93). Having obtained her first graduate degree in Solid State Physics at the University of Paris (1999), Tatjana was selected for the “Interface Physics-Biology” Graduate programme and after discovering a passion for developmental biology at the MBL Embryology Course in Woods Hole, she pursued a second PhD in Biology in the group of Sylvie Mazan at the University of Paris, with whom she worked to elucidate the conserved gene regulatory mechanisms of gastrulation (1-3). She then went on to work as a postdoctoral researcher in the group of Marianne Bronner at the California Institute of Technology. There, she pioneered experimental approaches to study gene regulation in vivo using avian embryos (4,5). In parallel, she developed methodologies to for the study the genomics of the sea lamprey, Petromyzon marinus (6,7). Tatjana spearheaded the establishment of a system in the Bronner lab to efficiently produce sea lamprey embryos throughout the summer, which became so successful that it still attracts researchers from all over the world for the “lamprey season” in Caltech. This enabled Tatjana to pioneer molecular biology approaches in lampreys, including loss-of-function experiments and the development of enhancer reporter assays. This early work laid the foundation for her subsequent contributions to illuminate the evolution of vertebrates, through the window of comparative studies of neural crest gene regulatory networks (reviewed in (8)).
In 2012 she became a group leader in the MRC Weatherall Institute of Molecular Medicine (WIMM) at the University of Oxford, where s
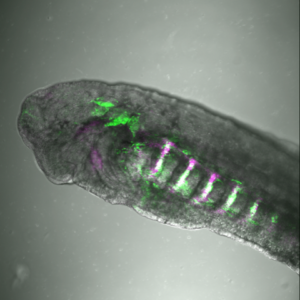
he was awarded the prestigious Lister Institute Research Prize (2013) and the March of Dimes Basil O’Connor Research Award (2013). Her keen interest in emerging technologies and the development of novel molecular biology tools allowed her to consistently stay at the forefront of regulatory genomics in developmental biology. Indeed, Tatjana pioneered enhancer screens to identify thousands of cell-type specific cis-regulatory elements in avian and zebrafish embryos, which together with single cell genomics allow for the reverse engineering of entire gene regulatory networks (9). Tatjana is extremely dedicated to carrying out her research meticulously and strives to broaden the range of technologies employed (i.e. Cut-and-Run- sequencing, machine learning approaches to next generation sequencing analysis) in order to break the boundaries of research in her field. Now an Associate Professor for Genome Biology at the University of Oxford generously supported by the Wellcome Trust Senior Research Fellowship (2019), the Sauka-Spengler lab uses the neural crest, the enteric nervous system and the zebrafish heart epicardium to explore the dynamics of gene regulatory networks in development, regeneration, disease and evolution (9-17).
What further distinguishes Tatjana as a Cheryll Tickle awardee is her passion and dedication to the fostering of young talent in the field developmental biology. This stretches beyond the mentoring of her own group, and breadth of support we have received in her nominations for the award is a true testament to her contributions to the developmental biology community in the UK. There is no better way to understand this than through the words of the many people who have benefitted from her knowledge and energy since establishing her lab in 2012. I’ll therefore leave you with the following quotes, that do so well to emphasize how deserving she is of this award.
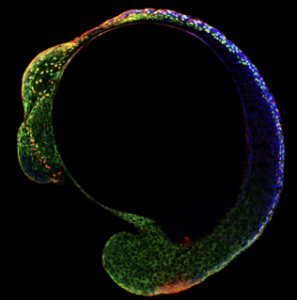
“As her mentees, we believe Tatjana’s unique mentorship style is her most outstanding characteristic, and the achievements she seems to be most proud of are those of the people she mentored. Indeed, her first graduate students, postdoctoral fellows and advisees are now establishing their labs (i.e., Betancur lab in UCSF, Hockman lab, Uni. of Cape Town, Simoes-Costa lab in Cornell Uni., Strobl-Mazzulla lab in IIB-INTECH, Argentina). Testament to this, in 2018 she was awarded the RDM Award for Excellent Supervision (University of Oxford)”. Chloe E. Tubman and Ivan L. Candido-Ferreira, DPhil candidates, University of Oxford.
“Tatjana is an inspirational role model for trainees and in particular for female scientists, for whom she provides guidance on balancing work and home life and inspires the confidence and direction to pursue individual goals”. Prof. Paul Riley, University of Oxford
“Simply put, Tatjana is an outstanding supervisor and mentor, not only to the people directly working with her, but also to any other junior scientist that approaches her for guidance”. Dr. Filipa Simões, University of Oxford.
“With her relentless energy and enthusiasm for excellent science combined with extensive knowledge and capacity to inspire, she has been a fantastic mentor. She sees the positive side of every situation and always provides a resolution, whether it be an experimental problem, writer’s block or personal matters”. Dr. Ruth Williams, University of Oxford.
“During our long-term collaboration Tatjana has hosted and supervised a number of my PhD students and post-doctoral fellows in her lab at the WIMM and trained them in maximising use of the zebrafish model to study heart development and regeneration. She has individually tutored my group members in bioinformatics to analyse RNA-Seq and ATAC-Seq datasets and modified gene editing approaches, which has been invaluable for the next stages of their research careers”. Prof. Paul Riley, University of Oxford.
“Tatjana very readily welcomes visiting scientists to her laboratory. She also generously shares her expertise and knowledge with collaborators. For example, she has applied her in vivo biotinylation approach in zebrafish (10,18), which enables the isolation of specific cell populations by affinity purification, to characterize the neutrophil response to mycobacterium infection (19). With colleagues she applied this approach to characterize different epicardial subpopulations (17), and has discovered how macrophages contribute to cardiac regeneration (16)”. Prof. Andrea Munsterberg, UEA
Ben Steventon,
Biography adapted from Chloe E. Tubman and Ivan L. Candido-Ferreira, DPhil candidates, University of Oxford. Additional input and editing provided by Tatjana Sauka-Spengler.
Selected papers:
- Sauka-Spengler, T., B. Baratte, M. Lepage, and S. Mazan, Characterization of Brachyury genes in the dogfish S. canicula and the lamprey L. fluviatilis. Insights into gastrulation in a chondrichthyan. Dev Biol, 2003. 263(2): p. 296-307.
- Sauka-Spengler, T., B. Baratte, L. Shi, and S. Mazan, Structure and expression of an Otx5-related gene in the dogfish Scyliorhinus canicula: evidence for a conserved role of Otx5 and Crxgenes in the specification of photoreceptors. Dev Genes Evol, 2001. 211(11): p. 533-44.
- Sauka-Spengler, T., A. Germot, D.L. Shi, and S. Mazan, Expression patterns of an Otx2 and an Otx5 orthologue in the urodele Pleurodeles waltl: implications on the evolutionary relationships between the balancers and cement gland in amphibians. Dev Genes Evol, 2002. 212(8): p. 380-7.
- Betancur, P., M. Bronner-Fraser, and T. Sauka-Spengler, Genomic code for Sox10 activation reveals a key regulatory enhancer for cranial neural crest. Proc Natl Acad Sci U S A, 2010. 107(8): p. 3570-5.
- Sauka-Spengler, T. and M. Barembaum, Gain- and loss-of-function approaches in the chick embryo. Methods Cell Biol, 2008. 87: p. 237-56.
- Nikitina, N., M. Bronner-Fraser, and T. Sauka-Spengler, The sea lamprey Petromyzon marinus: a model for evolutionary and developmental biology. Cold Spring Harb Protoc, 2009. 2009(1): p. pdb emo113.
- Sauka-Spengler, T., D. Meulemans, M. Jones, and M. Bronner-Fraser, Ancient evolutionary origin of the neural crest gene regulatory network. Dev Cell, 2007. 13(3): p. 405-20.
- Sauka-Spengler, T. and M. Bronner-Fraser, A gene regulatory network orchestrates neural crest formation. Nat Rev Mol Cell Biol, 2008. 9(7): p. 557-68.
- Williams, R.M., et al., Reconstruction of the Global Neural Crest Gene Regulatory Network In Vivo. Dev Cell, 2019. 51(2): p. 255-276 e7.
- Trinh, L.A., V. Chong-Morrison, D. Gavriouchkina, T. Hochgreb-Hagele, U. Senanayake, S.E. Fraser, and T. Sauka-Spengler, Biotagging of Specific Cell Populations in Zebrafish Reveals Gene Regulatory Logic Encoded in the Nuclear Transcriptome. Cell Rep, 2017. 19(2): p. 425-440.
- Williams, R.M., U. Senanayake, M. Artibani, G. Taylor, D. Wells, A.A. Ahmed, and T. Sauka-Spengler, Genome and epigenome engineering CRISPR toolkit for in vivo modulation of cis-regulatory interactions and gene expression in the chicken embryo. Development, 2018. 145(4).
- Kenyon, A., D. Gavriouchkina, J. Zorman, V. Chong-Morrison, G. Napolitani, V. Cerundolo, and T. Sauka-Spengler, Generation of a double binary transgenic zebrafish model to study myeloid gene regulation in response to oncogene activation in melanocytes. Dis Model Mech, 2018. 11(4).
- Lukoseviciute, M., et al., From Pioneer to Repressor: Bimodal foxd3 Activity Dynamically Remodels Neural Crest Regulatory Landscape In Vivo. Dev Cell, 2018. 47(5): p. 608-628 e6.
- Hockman, D., et al., A genome-wide assessment of the ancestral neural crest gene regulatory network. Nat Commun, 2019. 10(1): p. 4689.
- Ling, I.T.C. and T. Sauka-Spengler, Early chromatin shaping predetermines multipotent vagal neural crest into neural, neuronal and mesenchymal lineages. Nat Cell Biol, 2019. 21(12): p. 1504-1517.
- Simoes, F.C., et al., Macrophages directly contribute collagen to scar formation during zebrafish heart regeneration and mouse heart repair. Nat Commun, 2020. 11(1): p. 600.
- Weinberger, M., F.C. Simões, R. Patient, T. Sauka-Spengler*, and P.R. Riley*, Functional heterogeneity within the developing zebrafish epicardium. Dev Cell, 2020: p. doi: 10.1016/j.devcel.2020.01.023.
- Trinh, L.A., V. Chong-Morrison, and T. Sauka-Spengler, Biotagging, an in vivo biotinylation approach for cell-type specific subcellular profiling in zebrafish. Methods, 2018. 150: p. 24-31.
- Kenyon, A., D. Gavriouchkina, J. Zorman, G. Napolitani, V. Cerundolo, and T. Sauka-Spengler, Active nuclear transcriptome analysis reveals inflammasome-dependent mechanism for early neutrophil response to Mycobacterium marinum. Sci Rep, 2017. 7(1): p. 6505.